Abionic Early Sepsis Detection with Pancreatic Stone Protein (PSP)
Early Sepsis Detection with Pancreatic Stone Protein (PSP)
Sepsis
Sepsis is defined as life-threatening organ dysfunction caused by a dysregulated host response to infection.

Early recognition of sepsis is essential and is a major determinant of the disease’s outcome, according to The WHO and the Surviving Sepsis Campaign.
However this has proven to be challenging as the confirmation of sepsis diagnosis is based largely on nonspecific clinical signs, laboratory findings and medical scores which are usually obtained after sepsis onset.
Currently, C-reactive protein (CRP) and procalcitonin (PCT) are biomarkers routinely used for patients suspected of sepsis. Pancreatic stone protein or PSP is an emerging biomarker showing promise as it is characterized by higher accuracy in the diagnosis and prognosis of sepsis compared to CRP and PCT.
The Pancreatic Stone Protein (PSP)
• A 16 kDa polypeptide of the C-type lectin family of protein.
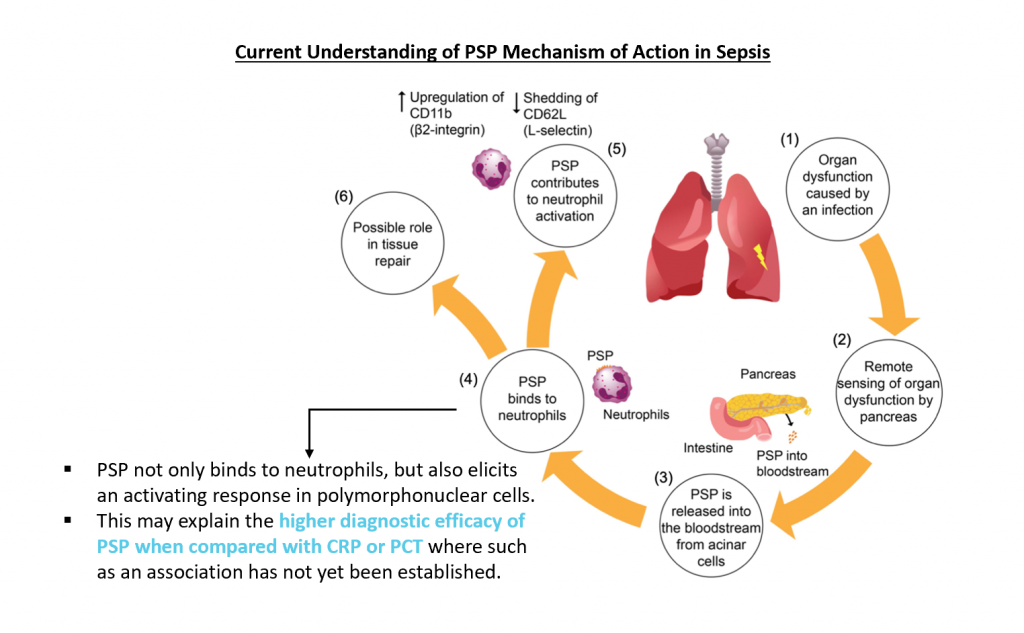
• Mostly secreted by the acinar cells of the pancreas but also by the intestine and stomach.
• An early sensor of sepsis and multiple organ dysfunction acting as an “alert signal” to help clinicians provide adequate infection control strategies and organ support to restore homeostasis.
Performance
PSP was proven to be more accurate, with a higher sensitivity, specificity, positive and negative predictive values than CRP, PCT, IL-6 and other cytokines for the diagnosis of sepsis and prognosis of unfavourable outcomes in multiple studies and clinical settings (ED, ICU, surgical, nonsurgical adult and children).
It also performed well in studies involving a variety of critically ill patients including severe burns, polytrauma, post-cardiac surgery and on admission to the ICU.
Unique Characteristic
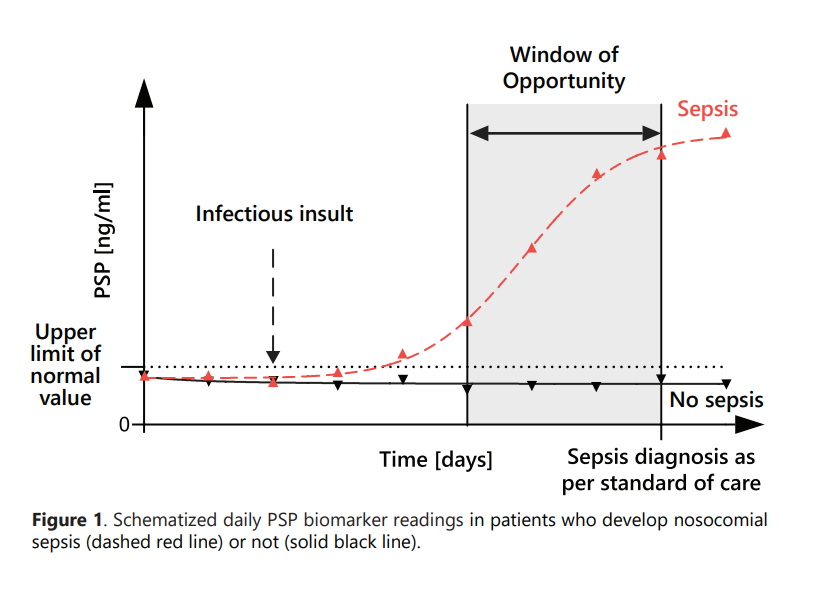
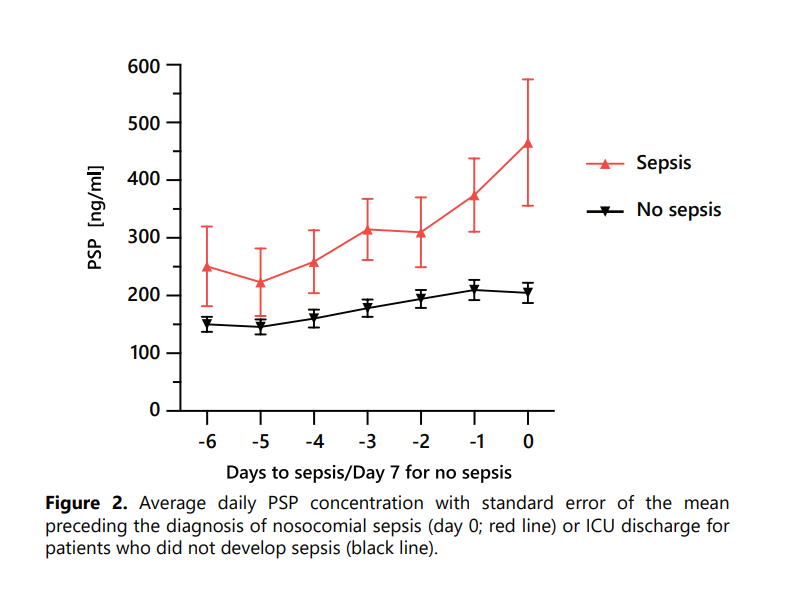
PSP may start to increase above the normal level before the development of clinical signs and symptoms of sepsis.
It was found to be the only biomarker able to identify sepsis 72 hours before clinical diagnosis, thereby providing a large window of opportunity for timely initiation of accurate clinical management.
PSP Measurement
Can now be done via a Point-of-Care(POC) device, which is uncommon for CRP and PCT in ICU, leading the way for simple, on-demand, around-the-clock, serial biomarker assessments instead of one-off testing upon clinical suspicion of sepsis.
This is key to faster treatment decisions, reducing mortality and lowering sepsis-related healthcare costs.
The IVD CAPSULE PSP on the abioSCOPE® is the first CE-marked in vitro diagnostic test to enable fast, reliable and early sepsis detection at the pointof-care from a single drop of blood in only 5 minutes.
Click the link below for more information:
Products | Abionic
References:
Eggimann, Philippe & Que, Yok-Ai & Rebeaud, Fabien. (2019). Measurement of pancreatic stone protein in the identification and management of sepsis. Biomarkers in Medicine. 13. 10.2217/bmm-2018-0194.
Pugin, J., Daix, T., Pagani, JL. et al. Serial measurement of pancreatic stone protein for the early detection of sepsis in intensive care unit patients: a prospective multicentric study. Crit Care 25, 151 (2021). https://doi.org/10.1186/s13054-021-03576-8
World Health Organization (WHO). Sepsis. https://www.who.int/news-room/fact-sheets/detail/sepsis
Rudd KE, Johnson SC, Agesa KM, Shackelford KA, Tsoi D, Kievlan DR, et al. Global, regional, and national sepsis incidence and mortality, 1990-2017: analysis for the Global Burden of Disease Study. Lancet (London, England). 2020;395(10219):200-11.
Klein HJ, Niggemann P, Buehler PK, Lehner F, Schweizer R, Rittirsch D, Fuchs N, Waldner M, Steiger P, Giovanoli P, Reding T, Graf R, Plock JA. Pancreatic Stone Protein Predicts Sepsis in Severely Burned Patients Irrespective of Trauma Severity: A Monocentric Observational Study. Ann Surg. 2021 Dec 1;274(6):e1179-e1186. doi: 10.1097/SLA.0000000000003784. PMID: 31972652.
Keel et al., 2009 and Reding et al., 2018 Pathophysiological mechanisms of PSP function in Sepsis- Abionic slides; Abionic brochure